-
I do not try to justify anything, I'm just trying to clarify that your oversimplification ("huge waste of energy") based on the fact that you lose 20x or 40x more over the RESISTIVE LOSES IN THE BATTERY is an exaggeration. Yea, sure if you're using a 1/10C charger for a 2Ah battery you're pissing away only 1mW "resistively", sure. And energy-wise you'll be using 40x more with a 4C charger. But it won't be 40 times the TOTAL losses, it will be 40x those tiny resistive loses. There are more loses in the battery itself (no, you don't get a charging efficiency of 99.5% with NiMh, even at 1/10C), losses in the charger, etc. You need to be looking at the whole package.
If you are looking at the whole package, you must also look for eficiency of the battery and you will conclude simply that it is better not to use batteries.
That's again a wrong way to analyse the situation.
If you are using batteries, it is because you need to do it.
But need you really charge your batteries in 15' at a tremendous high current rate with all the negative consequences this has ? (shortend live expectation, higher losses, very limited brands and types of batteries, even risk of explosion of the battery !)
Is it not better and more clever to buy several batteries more and to charge them at a lower rate ?
When you try to improve eficiency of a whole package, you have to improve eficiency of each part of this package.
1) battery charger: must be switch mode technology and be used in 25 - 100% output current range.
In this range, SMPS have a high eficiency.
You will say that my proposition of 3 charging currents (for 2 batteries, 8A, 2A and 0.4A) does not comply with this.
0.4A is only 5% of 8A.
But I never said that schematic of Varta is the good one.
I would have a different approach: the two converters principle, as used in computer SMPS and most of TV's sets and monitors: a low power auxiliary/stand by converter which would feed the µc and electronics control and also assume the 0.4A charging current of the batteries, and a power converter only switch on for charging with 2A or 8A rates.
2) Batteries: We can do nothing about battery technology and his efficiency, we can only reduce the losses during charging.
For this reason, never use the 8A current charging rate if it is not absolutely necessary.
I explained why but you don't seem to understand (or to accept !) what I wrote.
3) eficiency of what is powered by batteries: You seems to have forgotten this ! -
Another shortcoming of this Varta battery charger is the multiplexing when charging 4 batteries.
Why is this a shortcoming?
With unilateral pulsed current (here square wave), the charging current is the average current, but the current which cause Joule losses is the RMS current.
For the square wave, we have an average value of 1/2 the peak value and a RMS value of peak value/1.41 = 0.707 x peak current value.
Power losses in the batteries during charging are then 2x greater with a multiplexing that with a dc charging current. -
Quote from: PowerStream
The coulometric charging efficiency of nickel metal hydride batteries is typically 66%, meaning that you must put 150 amp hours into the battery for every 100 amp hours you get out. The faster you charge the worse this gets.
It is more like 90% to 95%, except the last part. -
Since resistive losses increase with the square of the current, I'm surprised the "4A" with 3 or 4 cells is implemented as a 50% duty cycle of 8A switched between the two banks. If they just did a constant 4A, it would theoretically cut the resistance losses in half.
Yes, but it would cost much more in the inductor of the DC/DC converter to allow for the higher dynamic in voltage
The additional loss per mosfet is in the order of 72mW, which is negligible, especially considering they have fan cooling
Actually if you assume 4.3momhs for the the mosfets (they'll be hotter than 25C) the extra loss is actually 206mW; add in the battery IR (25mohms), battery contacts (approx 1mohm each) and PCB traces (say 2mohms per cell) and the total extra loss for charging 4 batteries is 3.8W - not brilliant.
About those DTM4410 FETs - another bullshit datasheet. The datasheet says 3.8mohms typical, 4.5 max at 10V Vgs, 10A Ids - but the Rds(on) v Ids chart shows 7.8mohms typical at 10A, 10V - more than double! Same story for 4.5V Vgs . The Rds/Vgs chart also shows 7.8mohms at 10V, 10A at 10V, 10A so at least the two charts are consistent.
I had a look at several other Din-Tek mosfet specs and in almost all of them the Rds(on) charts bore no resemblance to the specs; e.g. the DTC2058 - spec is 26mohms, chart shows 15.7 (Vgs = 4.5V, Ids=8.3A). Many have Rds(on) scales which didn't make sense such as the attached chart for the DTL60N03 (specs are 3.1mohms @ 10V and 7.1millohms at 4.5V, both at 20A). You expect the odd error in datasheets but these seem to be mostly wrong - as a Japanese company I would have expected better.
So what do you trust? Presumably the DTM4410 3.8mohm typ, 6.5mohm max at 10A spec are the 'legal' figures but does that mean all the charts are worthless? Do they just make up the numbers or perhaps they have converted the graphs from pen and paper form using some dodgy OCR type software (surely not)?
-
Quote from: PowerStream
The coulometric charging efficiency of nickel metal hydride batteries is typically 66%, meaning that you must put 150 amp hours into the battery for every 100 amp hours you get out. The faster you charge the worse this gets.
It is more like 90% to 95%, except the last part.
No, NiMH is inherently much less efficient. This has to do with the difference between electrode potential and neutral charging voltage, which annoyingly both change throughout charging/discharging. They even change at the same charging setting and %DoD because the electrolyte depletes fairly easily. If you have a perfectly tracking charger at about 0.05-0.1C (i.e. one that stays on the exact right voltage to force the right current through the electrolyte and internal ohmic resistance), the best that can be attained is about 85%. Charging slower won't improve overall efficiency as you start running into significant self-discharging effects (due to the effect that higher electrolyte mobility during charging also induces higher self-discharge rates). In practice, chargers usually are constant-voltage with some dissipative element forcing constant-current; as well as charging at 0.5C, which is why they quote the 66% figure.
It's very important to note that NiMH is fundamentally different from li-ion in this respect. NiMH has very good ohmic internal resistance (basically on par with li-ion), but (relatively) bad ionic resistance. Li-ion has incredibly low ionic resistance, which means that charging and discharging is basically only limited by the resistance of the electrodes. This is why efficiency is not an issue with li-ion, and li-ion can sustain much higher currents without nearly as much impact on capacity. -
Quote from: PowerStream
The coulometric charging efficiency of nickel metal hydride batteries is typically 66%, meaning that you must put 150 amp hours into the battery for every 100 amp hours you get out. The faster you charge the worse this gets.
It is more like 90% to 95%, except the last part.
No, NiMH is inherently much less efficient. This has to do with the difference between electrode potential and neutral charging voltage, which annoyingly both change throughout charging/discharging.
Instead of writing theories, try measuring it, I did.
-
Instead of writing theories, try measuring it, I did.
Maybe you should take that up with PowerStream, since you apparently know far more about batteries than they do. It's not like it's their core business or anything. -
Instead of writing theories, try measuring it, I did.
Maybe you should take that up with PowerStream, since you apparently know far more about batteries than they do. It's not like it's their core business or anything.
They and you can just read on my website, where this will be published, together with all my other battery stuff. You could also do you own testing and learn something, instead of just believing a random article. -
Why is there even an argument about the energy efficiency of the charger, no-one is going to buy a 15 min NiMh charger based on the efficiency!
What matters is if it works and the economy of it, i.e. how long the batteries are going to last (the electricity cost is going to be negligible unless there is some large standby current and it's always plugged in, but that would not be because it's a 15-minute charger). I think most realize there will be a compromise between battery life and charge current. I agree its not a good solution technically if you can plan ahead and manage your batteries properly, but most people know nothing about batteries and they don't have enough foresight to charge the batteries in advance. I remember being a kid and never having fresh batteries. Charging them would take a day so it effectively killed whatever activity you needed them for (and my parents were too cheap (or devious) to have enough spares that could be charged in advance). So in short: a 15 minute charger would have been awesome. The same must be true today, if someone picks up a game controller (or whatever) and realize the batteries are flat, waiting 15 minutes is not so bad, 2 hours (or 15 hours for trickle charging) is going to be a show stopper. -
Instead of writing theories, try measuring it, I did.
Maybe you should take that up with PowerStream, since you apparently know far more about batteries than they do. It's not like it's their core business or anything.
They and you can just read on my website, where this will be published, together with all my other battery stuff. You could also do you own testing and learn something, instead of just believing a random article.
Do you have a link? -
Do you have a link?
Here: http://lygte-info.dk/
Mostly it is technical reviews, but once in a while I publish some articles. You can find them by careful looking through the battery & charger section.
The one about NiMH charging is not published yet. -
@apis:Quote
Why is there even an argument about the energy efficiency of the charger, no-one is going to buy a 15 min NiMh charger based on the efficiency!
Eficiency is a major concern in the modern world.
Perhaps electric energy is very cheap in Sweden, that's not true in other lands.Quote(and my parents were too cheap (or devious) to have enough spares that could be charged in advance)
For sure, your parents would also buy low cost batteries who would never accept to be charged with 4C.
Charging at 4C is above specifications of every AA batteries manufacturer and, if I remember right, Dave has showed that even Varta don't allow such a high rate with his batteries.
Using such a tremendous high rate has a high cost:
1) only high quality batteries can be used.
2) life expectancy is reduced.
3) losses are higher. -
I did not seen any efficiency value of batteries...Instead of writing theories, try measuring it, I did.
Maybe you should take that up with PowerStream, since you apparently know far more about batteries than they do. It's not like it's their core business or anything.
They and you can just read on my website, where this will be published, together with all my other battery stuff. You could also do you own testing and learn something, instead of just believing a random article.
What efficiency are you speaking about ?
Faraday or energetic efficiency ?
Energetic efficiency is lower than Faraday one.
How will you measure and calculate energetic efficiency of a AA battery ?
Easiest way would be to charge and discharge the battery at constant power and measure the times of charge and discharge.
You can discharge the battery at constant power with your electronic load, but how are you charging the battery with constant power ?
Your power supply does not have this option. -
I did not seen any efficiency value of batteries...
What efficiency are you speaking about ?
Faraday or energetic efficiency ?
Energetic efficiency is lower than Faraday one.
How will you measure and calculate energetic efficiency of a AA battery ?
Easiest way would be to charge and discharge the battery at constant power and measure the times of charge and discharge.
You can discharge the battery at constant power with your electronic load, but how are you charging the battery with constant power ?
Your power supply does not have this option.
I have not published the article yet. The test is for mAh in and mAh out, where I uses a 2000mAh cell and test it with values from 1500mAh to 2300mAh. -
Ok, that's the Faraday efficiency, that's the reason why your values are so high.
The true efficiency is the energetic efficiency, that's how much energy in and how much energy out. (energy = power x time. If power varies during the time of charge or discharge, you must integrate the power during this time)
I suggest you to measure this efficiency and not the Faraday efficiency who does not mean nothing ! -
Ok, that's the Faraday efficiency, that's the reason why your values are so high.
The true efficiency is the energetic efficiency, that's how much energy in and how much energy out.
I suggest you to measure this efficiency and not the Faraday efficiency who does not mean nothing !
The statement I answered was this "meaning that you must put 150 amp hours into the battery for every 100 amp hours you get out" and that is not correct.
I do have the voltages during charge and discharge and can calculate the energy efficiency, if I can find the time I may do it.
-
Instead of writing theories, try measuring it, I did.
Maybe you should take that up with PowerStream, since you apparently know far more about batteries than they do. It's not like it's their core business or anything.
They and you can just read on my website, where this will be published, together with all my other battery stuff. You could also do you own testing and learn something, instead of just believing a random article.
I suspect your test methodology was incorrect then, though as we can't see it we can't tell. Coulombic efficiency can be high if you only partly charge the cells, but rapidly drops as you approach full capacity and 66% is a typical figure for a fully charged NiMh cell. -
I suspect your test methodology was incorrect then, though as we can't see it we can't tell.
As I have already explained that I charged different mAh into the cell and the discharged it again to measure the mAhCoulombic efficiency can be high if you only partly charge the cells, but rapidly drops as you approach full capacity and 66% is a typical figure for a fully charged NiMh cell.
That is correct mostly correct, with the 2000mAh cell the efficiency was high up to about 1900mAh into the cell (>1850mAh out). If you insist on filling 2500mAh into it you will get a bad efficiency. -
@HKJ:Quote
I do have the voltages during charge and discharge and can calculate the energy efficiency, if I can find the time I may do it.
Calculate ? I wonder how you could do this as voltages are varying during charge and discharge in fonction of the time at a non linear rate ?
Could you explain how should you calculate the energetic efficiency ? What are the formulas you intend to use ? -
@HKJ:
QuoteI do have the voltages during charge and discharge and can calculate the energy efficiency, if I can find the time I may do it.
Calculate ? I wonder how you could do this as voltages are varying during charge and discharge in fonction of the time at a non linear rate ?
Could you explain how should you calculate the energetic efficiency ? What are the formulas you intend to use ?
I have log points for each second, it is easy to calculate power for each log point and then sum power*time. This would give my total charged and discharged energy.
My logfile looks like this (i.e. I have already done the calculations, I just need to collect them from a couple of files):
time SMMeasure SMSource SMOhm SMW SMAh SMWh
0 1,028179 0,99999 1,028189 1,028169 0,000257 0,000264
1 1,044294 0,999991 1,044303 1,044285 0,000535 0,000555
2 1,061145 0,999992 1,061154 1,061136 0,000813 0,000849
3 1,078734 0,999991 1,078744 1,078724 0,001091 0,001149
4 1,096882 0,999992 1,096891 1,096873 0,001368 0,001454
5 1,115237 0,999992 1,115246 1,115228 0,001646 0,001763
-
That's an approximation, but it is ok.
Have you already make this calculation for one kind of battery ? (as eneloop for example).
I am curious to see wat you find as energetic efficiency. I have in mind something like 80 to 90% but I did not measure this. -
That's an approximation, but it is ok.
It will be difficult to do it any better. With a real cell you have to measure in discrete time intervals. Converting the data to a smooth curve may give slightly better precision.Have you already make this calculation for one kind of battery ? (as eneloop for example).
I have the data for a single NiMH cell at different charge capacities, but have not picked them out of the log files.
I am curious to see wat you find as energetic efficiency. I have in mind something like 80 to 90% but I did not measure this. -
I hope you will share the results of your compiled data, it will be very interesting, you have done a very fine job

-
I hope you will share the results of your compiled data, it will be very interesting, you have done a very fine job

Here you have the curve with energy: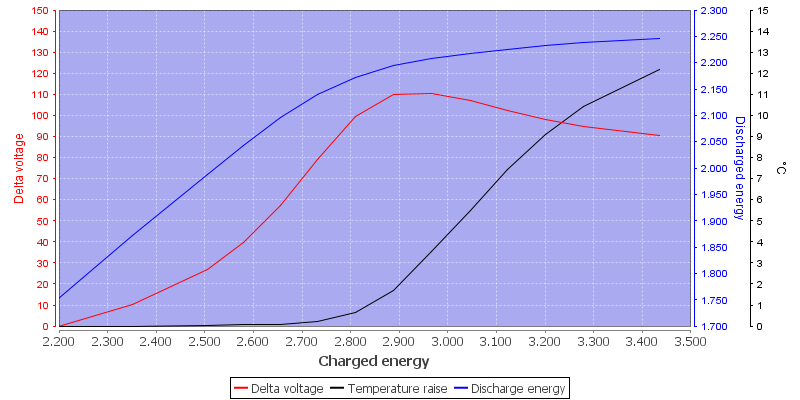
Delta voltage is voltage above 1.50 volt
At best you get 80% of the energy back.
For the rest of the article see here: http://lygte-info.dk/info/batteryChargingNiMH%20UK.html
The mAh efficiency is just a small part of the article. -
Has anyone figured out what processor is used in this charger yet?
I had a quick look at the voltages on the pins, and most are either 0 or 5V, indicating a 5V processor. But one pin puzzled me: Pin 18 is at 5.65V
They could be feeding it higher than Vcc through a resistor, and relying on the processors internal esd protection diode to clamp the voltage.