What is really incredible, is that they plan to release this stuff in november, and there are at least 5 or 6 different version of this that has been show, the copperish one, the one with pink, but we can see more different design on the videos they show..
I suspected they already had had them manufactured before they began the gogo campaign, since the prototypes in the videos look rather polished, but I'm not so sure any more since they appear to continuously update the design based on feedback from dave and this forum.

In one of their videos they show someone putting them in an Apple wireless keyboard, with the typical "battery tube" end-loader. I'd much prefer to see them getting the batteries back out of it, that would be a much more entertaining video!

McBryce.
Hi group,
the 800% explanation.
This is from the Batteriser Patent Application.
If one looks at the potential return of such a device in terms of lifetime of a battery, one can see significant benefits. For instance, the AA battery in the above example would use roughly the equivalent charge of the battery output in the range of 1.5V to 1.4V. This means that after 0.1V drop, the battery's life is over. If the battery could be used until its voltage reaches 0.8V, then after 0.7V drop the battery's life is over. If one were to assume that the time versus the voltage drop is a linear function, then the life of the battery could be improved by a factor of 7 in this example. However, advantageously the time versus voltage drop is not quite linear. The time it takes for the battery voltage to drop by 0.1V is longer at lower voltages versus at higher voltages. That means that if a constant current was drawn from the battery, it would take the battery a lot longer to discharge from 1.2V to 1.1V than it would from 1.5V to 1.4V. This means that the extent to which the battery life is increased could be even higher than the factor of 7 in the above example above.
It is noted that the regulation circuit has a certain efficiency which cuts back on the extent to which the battery life is extended though the life time reduction is rather minimal. During operation, the regulator itself uses a certain amount of current from the battery. A lot of the available DC to DC converters have high efficiencies of around 95%. That is, of power supplied by the battery, 5% is used by the converter and the rest is available for the end user. However, the 5% efficiency loss due to use of a converter, when compared to the 700% gain in efficiency of the battery, is negligible. It is further noted, that the converter efficiency may drop as the battery voltage drops due to use. For example, as the battery voltage drops from 1.5V to 1V, the efficiency of the converter may drop down to 50% to 60%. However, 50% efficiency is still a significant improvement over the current approach of discarding the batteries because their voltage has dropped below the operable voltage range (i.e., 1.4-1.5V).
Since they talk about the battery having a linear discharge curve, we can treat this as a capacitor:
Assume a AA battery is 2Ahr the capacitance is 3600x2 = 7200F
I=C dv/dt
Assume the powered device has a cut-off voltage of 1.4, the voltage has to fall 0.1V
Assume a 50mA power consumption
solving for t
The device runs for 14400 seconds.
Now will introduce a lossless Boost converter.
The properties of a boost converter are power is preserved, the power consumption from the battery will be constant,
The power consumption is 1.5V x 50mA = 75mW
If the Starting voltage is Vo
And the end point voltage is V1
The discharge time can be calculated as
t = 0.5 x C (V02 - V12) / power consumption
If I plug in the numbers I get
T= 0.5 x 7200 (1.52 - 0.72)/75mW = 84,480 seconds
Even in this idealised case the time multiplier is
84,480 / 14400 = 5.8x
If the cut-off is 1.3V the multiplier is x0.5 = 2.9x
It seems like Dr. Bob didn't get the math right in the patent application !!
Adding inefficiencies, or raising the UVLO for the boost circuit only makes this worse not better.
Of course the battery discharge is not a linear function of time, it has a plateau in it. Any product that is designed (correctly) until after the plateau will work longer without the batteriser than with it.
Jay_Diddy_B
Jay_Diddy_B,
I don't know where you find the time to devote to disproving the batteriser claims. Nonetheless, good job. The scientific community requires proof for a claim, however, it seems, rather sadly, that the consumer market accepts a claim blindly and requires someone to DISPROVE it. If more people paid attention to the EEVblog, I don't think they would even spend their time laughing at their silly claims.
They answered one of my question on Youtube, see the "Batteriser - Full length Batteriser explanation video" video. The answer is a bit confusing, and with obvious errors in it ("Y axis is hours"). Another one answered to their answer, they deleted it. I found the text still in my eMail notification:
gblargg: +Batteriser Batteroo From Wikipedia, you need to include the voltage to determine power remaining in the battery: "An ampere-hour is not a unit of energy. In a battery system, for example, accurate calculation of the energy delivered requires integration of the power delivered (product of instantaneous voltage and instantaneous current) over the discharge interval. Generally, the battery voltage varies during discharge; an average value or nominal value may be used to approximate the integration of power."?
The text from wikipedia is from
this article. But
this Wikipedia article says "Capacity is measured in units such as amp-hour (A·h)" and this is what I can see on my batteries here. Before I answer, is it right to measure the capacity in mAh? What terminal voltage is used for it? I know physically it is not accurate, because as the Wikipedia ampere-hour article says, you have to integrate it over time with the current voltage and current. Would be better if the batteries would specify the capacity in Wh.
The text from wikipedia is from this article. But this Wikipedia article says "Capacity is measured in units such as amp-hour (A·h)" and this is what I can see on my batteries here. Before I answer, is it right to measure the capacity in mAh?
No it's not right, and anyone who does any serious work with battery capacity and energy knows that.
But, it is common to simply (incorrectly) use mAh, as it's actually generally not that far off as evidenced by the almost linear capacity curve in my blog post and video here:
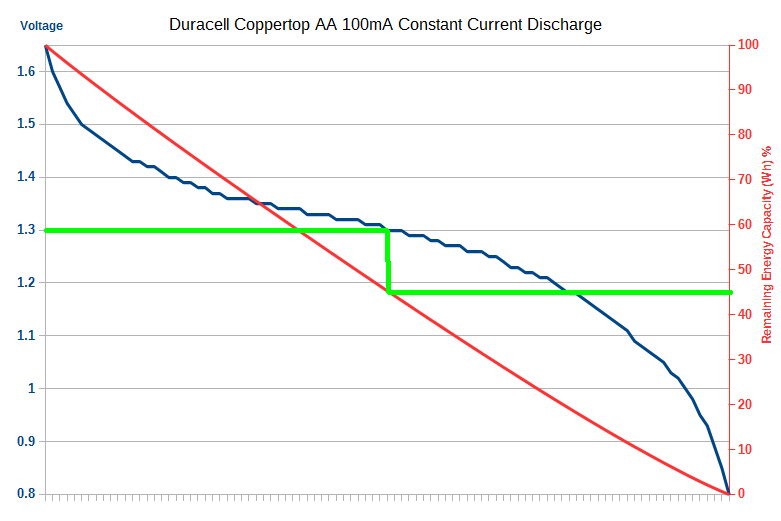
If everyone uses mAh then it's an acceptable comparison metric.
What terminal voltage is used for it? I know physically it is not accurate, because as the Wikipedia ampere-hour article says, you have to integrate it over time with the current voltage and current. Would be better if the batteries would specify the capacity in Wh.
Wh is common on mobile phone and laptop batteries, because of confusion with multi-cell packs of different voltages.
Wh is the only true measure of capacity, and it's no different for single Alkaline cells.
EEVBlog forum points out a serious shortcoming. Batteroo makes a change shortly thereafter. Repeat. It may all be a series of incredibly remarkable coincidences. 
Only a dozen times in a row now. Yeah, must be coincidence.

It's so great that the EEvblog has been so instrumental in the Batteriser's development. I wonder if there will ever be attribution, or at least a simple thank you.
It seems that no one on the forum, including myself, have been offered sample units to test.
You'd think they could get no better technical feedback (and real glowing reviews proving it really works) than from those on here

If I remember correctly a few year year ago, it has been decided that battery capacity must be displayed in Wh instead of the more commonly used (m)Ah due to some international regulation. It may only touch Lithium based battery I'm not sure.
Does anyone knows more about that?
If I remember correctly a few year year ago, it has been decided that battery capacity must be displayed in Wh instead of the more commonly used (m)Ah due to some international regulation. It may only touch Lithium based battery I'm not sure.
Does anyone knows more about that?
The batteries themselves might, but that doesn't stop manufacturers from using Ah instead to make their capacity look larger.
Best example: USB power banks. It drives me insane when I see ads saying that a 10,000 mAh USB power bank can charge a 2,000 mAh smartphone five times. Little do they know, that the 10,000 mAh rating is at the 3.7 volts of the lithium cells inside the unit (all in parallel), and the mAh that you get out of the unit is much less when you convert to Wh, then back to mAh @ 5v that it gets boosted to by the boost converter. (not to mention the losses)
Marketing marketing marketing
is it right to measure the capacity in mAh? What terminal voltage is used for it? I know physically it is not accurate, because as the Wikipedia ampere-hour article says, you have to integrate it over time with the current voltage and current. Would be better if the batteries would specify the capacity in Wh.
'Better' depends on the device. mAh is a more useful metric for a constant current device, Wh is more useful for a constant power device.
In reality, neither can be used to sit down and calculate exactly how long a battery is going to last because there's other factors, eg. temperature.
The main point of battery markings is to be able to compare batteries with each other. mAh works for that.
The car analogy would be "mpg"*. It lets you compare cars with each other but everybody knows that you won't get that fuel consumption in practice.
(Or l/100km, depending on where you live)
Another Batteriser rendering now.
Completely busting the monkey video, with the real monkey.
Yes, lots of waffle

Another Batteriser rendering now.
Completely busting the monkey video, with the real monkey.
Yes, lots of waffle 
I'm looking forward to watching you play with your monkey

Another Batteriser rendering now.
Completely busting the monkey video, with the real monkey.
Yes, lots of waffle 
Dave, tell us the truth; you really like to do theses videos

Just saw this...
/?action=dlattach;attach=168473;image)
Where on earth could anyone possibly get that idea from!?!


Just saw this...
/?action=dlattach;attach=168473;image)
Where on earth could anyone possibly get that idea from!?!


He answer to the BB question should be
"In one monkey butt"
* Godzil is searching for a door...
Just saw this...

No, no, no...
The comedy is how this (original response):
/?action=dlattach;attach=168481;image)
Became this (what it says today):

There must have been a real "Oh, shit!" moment between those two posts.
Still, credit where credit is due: He
did manage to put a compliance "team" together in less than 24 hours. On a weekend.

They will also need to comply with the EU "CE" requirements for the EMC directive.
I love that Troll there............

I have a Crompton TCL lithium primary cell that is dead, but it still has 3V7 into 10M, but internal resistance is in the tens of kilohms range. Expired a decade ago, but still good enough to run a CMOS memory which uses less than 1uA in standby.
Latest video busting that poor Monkey:
Where did you get the troll?

















